ultrasound guided sclerotherapy
Ultrasound-Guided Sclerotherapy: A Comprehensive Overview (as of 03/05/2026)
Ultrasound-guided foam sclerotherapy (UGFS) effectively treats varicose veins, proving a safe option for smaller, limited cases․ Current evidence supports its efficacy and safety․
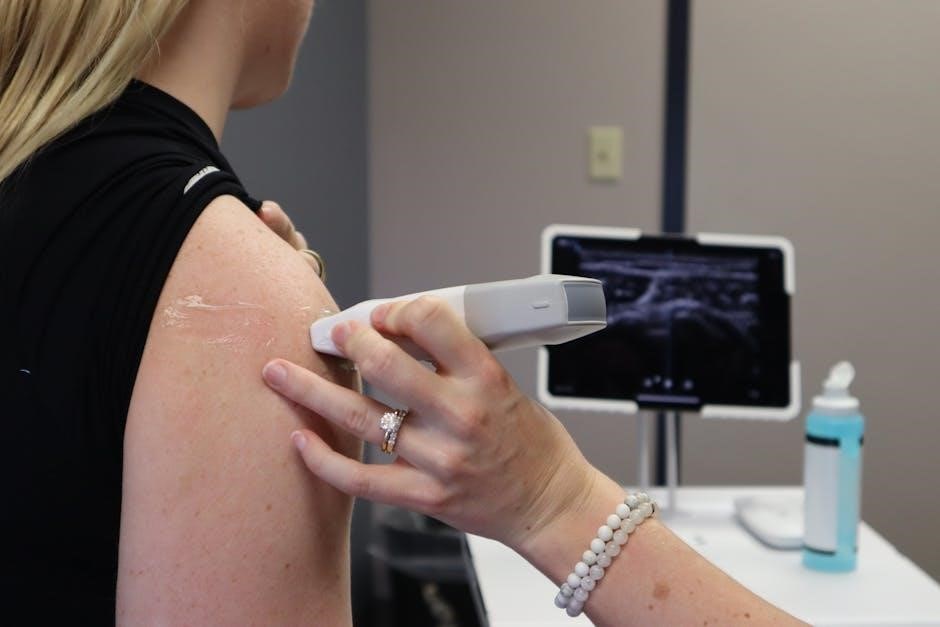
Ultrasound-Guided Sclerotherapy (UGFS) represents a significant advancement in the treatment of varicose veins, combining the established technique of sclerotherapy with real-time ultrasound guidance․ Traditionally, sclerotherapy involved injecting a solution directly into the vein, relying on palpation to ensure accurate placement․ However, UGFS utilizes ultrasound imaging to visualize the vein, allowing for precise injection of the sclerosing agent, particularly beneficial for smaller or more complex varicose veins․
This enhanced precision improves treatment efficacy and minimizes the risk of complications․ As of today, March 5th, 2026, UGFS is recognized as a useful treatment option, especially for limited varicose veins․ The migration of Interventional Procedures Guidance 440 to HealthTech Guidance 301 underscores its established place within clinical practice, with recommendations remaining consistent․ UGFS offers a relatively safe and effective alternative to more invasive procedures․
What are Varicose Veins and Why Treat Them?
Varicose veins are enlarged, twisted veins that typically appear close to the skin’s surface, most commonly in the legs and feet․ They develop when the valves within veins become weakened or damaged, leading to blood pooling and increased pressure․ This results in the characteristic bulging appearance and can cause discomfort, including aching, heaviness, and swelling․
Treatment isn’t solely cosmetic; addressing varicose veins can significantly improve quality of life․ While ultrasound-guided sclerotherapy (UGFS) is a key treatment, understanding the underlying condition is crucial․ Untreated varicose veins can progress, leading to complications like skin ulcers, superficial thrombophlebitis, and, rarely, deep vein thrombosis․ UGFS, as a relatively safe method for limited veins, offers a targeted approach to alleviate symptoms and prevent these potential issues, improving venous circulation and overall well-being․
The Role of Ultrasound in Sclerotherapy
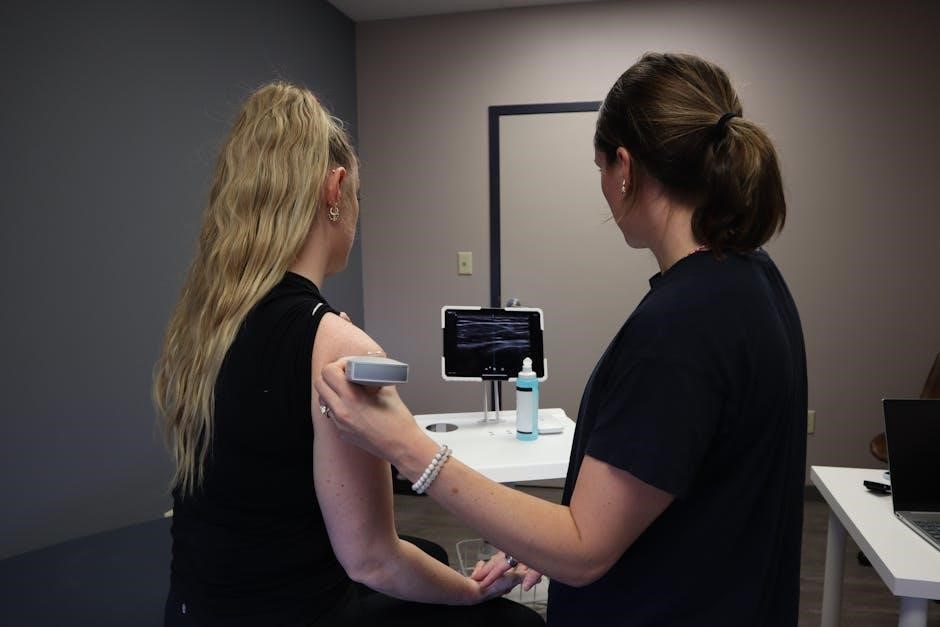
Traditional sclerotherapy, while effective, benefits immensely from the integration of ultrasound guidance, transforming it into Ultrasound-Guided Sclerotherapy (UGFS)․ Clinical ultrasound is invaluable, not only for detecting broader vascular issues like atherosclerosis, but also for precise visualization of varicose veins․ This real-time imaging allows practitioners to accurately assess the vein’s size, location, and any connecting branches – crucial for targeted treatment․

UGFS ensures the sclerosing agent is delivered directly into the problematic vein, minimizing exposure to surrounding tissues and maximizing efficacy․ It also aids in confirming complete vein collapse during and after the procedure․ The lack of standardized methods for assessing vascular areas with plaques highlights the importance of UGFS’s precision, offering a more consistent and reliable approach to varicose vein treatment․
Procedure Details
UGFS involves careful preparation, precise sclerosing agent selection, and a step-by-step technique utilizing ultrasound for guidance, differentiating between foam and liquid sclerotherapy approaches․
Preparation for UGFS
Prior to undergoing Ultrasound-Guided Sclerotherapy (UGFS), a comprehensive evaluation is crucial․ This begins with a detailed medical history review, focusing on any pre-existing conditions, allergies, and current medications – particularly anticoagulants․ A thorough physical examination of the affected veins is then performed to assess their size, location, and overall condition․
Patients are typically advised to avoid certain medications, like aspirin or NSAIDs, for a specified period before the procedure to minimize the risk of bleeding․ It’s also recommended to discontinue any topical creams or lotions on the treatment area․ On the day of the procedure, patients should arrive with comfortable clothing and may be asked to avoid heavy meals beforehand․
The interventional radiologist or phlebologist will clearly explain the procedure, potential risks, and benefits, ensuring the patient has a complete understanding and can provide informed consent․ Pre-procedural ultrasound imaging is often conducted to precisely map the varicose veins and guide the sclerosing agent delivery․
The Sclerosing Agent: Types and Selection
The cornerstone of UGFS lies in the sclerosing agent, substances inducing vein wall inflammation and eventual closure․ Commonly used agents include sodium tetradecyl sulfate (STS), polidocanol, and sodium chloride․ STS is frequently favored for its proven efficacy, while polidocanol offers a lower risk of skin pigmentation․ The choice hinges on vein size, patient history, and physician preference․
Sclerosing agents are available in liquid and foam formulations․ Foam sclerotherapy, utilizing a gas mixed with the liquid agent, provides prolonged contact with the vein wall, enhancing effectiveness, particularly for larger varicose veins․ Agent concentration is carefully adjusted based on vein diameter; higher concentrations are used for larger veins․
Prior to injection, the agent is thoroughly inspected for particulate matter․ Patient allergies and sensitivities are meticulously considered during selection, ensuring a safe and tailored treatment approach․
UGFS Technique: Step-by-Step Guide
The UGFS procedure begins with the patient positioned supine, allowing optimal ultrasound visualization․ After thorough skin disinfection, a small needle is inserted into the targeted varicose vein under real-time ultrasound guidance․ Correct needle placement is crucial, avoiding adjacent structures․
Following successful access, a small amount of the sclerosing agent – either liquid or foam – is slowly injected into the vein․ Ultrasound confirms proper agent distribution within the vein, ensuring complete coverage․ The injection is performed incrementally, monitoring for any immediate adverse reactions․
Post-injection, gentle pressure is applied to the puncture site․ The process is repeated for other identified varicose veins within the treatment area, systematically addressing the venous insufficiency․ Throughout, continuous ultrasound monitoring guides the physician, maximizing precision and safety․
Foam Sclerotherapy vs․ Liquid Sclerotherapy
Both foam and liquid sclerotherapy aim to obliterate varicose veins, but differ in their approach․ Liquid sclerotherapy utilizes a solution directly injected into the vein, relying on chemical irritation for closure․ It’s often suitable for smaller veins and spider veins․
Foam sclerotherapy, however, mixes the sclerosing agent with air, creating a foam․ This foam fills the vein more completely, particularly beneficial for larger, more complex varicose veins․ The foam displaces blood, enhancing contact with the vein wall․
UGFS frequently employs foam sclerotherapy due to its superior efficacy in larger veins, confirmed by ultrasound visualization of complete vein filling․ While both methods are generally safe, foam may offer improved outcomes and reduced recurrence rates in specific cases, guided by careful patient assessment․
Benefits and Risks
UGFS demonstrates adequate efficacy and a generally safe profile, though patients must be informed of potential, albeit small, risks associated with the procedure․
Efficacy of UGFS for Varicose Veins
Current evidence strongly suggests that ultrasound-guided foam sclerotherapy (UGFS) is an adequate and valuable treatment modality for addressing varicose veins․ The technique’s efficacy is particularly notable when applied to limited and smaller varicose vein presentations․ The guidance, now transitioned from Interventional Procedures Guidance 440 to HealthTech Guidance 301, confirms this positive assessment, with recommendations remaining unchanged․
UGFS achieves successful vein closure through the targeted delivery of a sclerosing agent, visualized and guided by real-time ultrasound imaging․ This precision enhances treatment outcomes and minimizes potential complications․ While clinical ultrasound excels in detecting broader vascular issues like atherosclerosis, UGFS specifically focuses on the targeted treatment of problematic varicose veins, offering a focused and effective solution for patients seeking relief and improved cosmetic results․ The consistent positive findings support its continued use as a reliable treatment option․
Safety Profile of UGFS: Common and Rare Complications
Ultrasound-guided foam sclerotherapy (UGFS) is generally considered a relatively safe procedure for treating varicose veins, particularly in cases involving limited, smaller veins․ However, as with any medical intervention, potential complications exist․ The current guidance (HealthTech Guidance 301) emphasizes the importance of adequately warning patients about these possibilities, even though they are typically small in occurrence․
Common, minor complications may include temporary bruising, swelling, or discomfort at the injection site․ These are usually self-limiting and resolve within a few days or weeks․ Rarer, more serious complications, while infrequent, can include skin necrosis, allergic reactions to the sclerosing agent, or, very rarely, deep vein thrombosis․ Thorough patient assessment and adherence to established protocols are crucial to minimizing these risks․ Careful monitoring post-procedure is also essential for early detection and management of any adverse events․
Patient Selection Criteria for UGFS
Determining appropriate candidates for Ultrasound-Guided Foam Sclerotherapy (UGFS) is crucial for optimal outcomes and minimizing risks․ Generally, UGFS is best suited for patients with isolated, smaller varicose veins․ HealthTech Guidance 301 supports its use in these specific cases․
Ideal patients typically present with symptomatic varicose veins that haven’t responded to conservative management like compression stockings․ A thorough vascular assessment, including ultrasound, is essential to rule out underlying venous insufficiency or deep vein thrombosis․ Patients with significant co-morbidities, such as severe cardiovascular disease, may not be suitable candidates․ Careful consideration is given to skin condition, as compromised skin increases the risk of complications․ Ultimately, a personalized approach, weighing the benefits against potential risks, guides patient selection for UGFS․

Post-Procedure Care and Follow-Up
Post-UGFS, compression therapy is vital․ Managing discomfort is key, alongside follow-up ultrasound assessments to monitor treated veins and ensure successful outcomes․
Compression Therapy After UGFS
Following Ultrasound-Guided Foam Sclerotherapy (UGFS), consistent compression therapy is paramount for optimal results and minimizing potential complications․ Graduated compression stockings are typically recommended, usually worn for a specified duration – often several weeks, sometimes extending to months – as directed by the treating physician․
The purpose of compression is multifaceted․ It aids in reducing post-procedural swelling and discomfort, promotes venous closure by increasing pressure within the treated veins, and helps prevent the recurrence of varicose veins․ The level of compression prescribed will vary based on individual patient needs and the extent of the UGFS treatment performed․
Patients should be thoroughly instructed on the proper application and wear schedule of compression stockings․ It’s crucial to ensure they are correctly sized and fitted to avoid constriction or discomfort․ Adherence to the prescribed compression regimen significantly contributes to the long-term success of the UGFS procedure․
Managing Post-Procedure Discomfort
Post-UGFS discomfort is common, typically manifesting as mild aching, bruising, or a feeling of heaviness in the treated legs․ These symptoms are generally self-limiting and resolve within a few days to weeks․ Over-the-counter pain relievers, such as paracetamol or ibuprofen, can effectively manage any discomfort, but aspirin should be avoided due to its potential to increase bleeding risk․
Applying cool compresses to the treated area during the first 24-48 hours can help minimize bruising and swelling․ Gentle walking is encouraged to promote circulation and reduce the risk of deep vein thrombosis (DVT), but strenuous activity should be avoided․
Patients should be advised to contact their physician if they experience severe pain, signs of infection (redness, warmth, pus), or symptoms suggestive of DVT (significant swelling, pain, tenderness)․ Prompt attention to any concerning symptoms ensures optimal recovery․
Follow-Up Ultrasound Assessments
Following Ultrasound-Guided Sclerotherapy (UGFS), routine follow-up ultrasound assessments are crucial to evaluate treatment efficacy and detect any potential complications․ A primary ultrasound is typically scheduled 6-12 weeks post-procedure to assess vein closure and identify any residual varicose veins requiring further treatment․
These assessments allow physicians to visualize the treated veins, confirm complete obliteration, and rule out the development of new venous reflux․ The timing of follow-up may vary based on individual patient factors and the extent of the initial treatment․
Periodic ultrasound surveillance can also help identify any late recurrence of varicose veins, enabling timely intervention․ Consistent monitoring ensures long-term success and minimizes the likelihood of symptom relapse, optimizing patient outcomes after UGFS․
UGFS and Other Treatment Options
UGFS presents a valuable alternative to traditional sclerotherapy and Endovenous Laser Ablation (EVLA), offering tailored treatment approaches based on vein characteristics and patient needs․
UGFS Compared to Traditional Sclerotherapy
Traditional sclerotherapy, performed without real-time imaging, relies on palpation to guide the injection of the sclerosing agent․ While effective for smaller varicose veins, it can be less precise for larger or more complex cases․ Ultrasound guidance in UGFS significantly enhances accuracy, allowing for visualization of the vein, surrounding structures, and precise delivery of the sclerosant․
This improved precision translates to several advantages․ UGFS minimizes the risk of injecting the sclerosant into unwanted tissues, reducing post-procedure complications like skin staining or inflammation․ Furthermore, ultrasound allows for the treatment of veins that are difficult to access or visualize through palpation alone․
UGFS also facilitates the use of foam sclerotherapy, which is often preferred for larger veins due to its increased contact time with the vessel wall․ Traditional sclerotherapy typically utilizes liquid sclerosants․ Ultimately, UGFS offers a more targeted and potentially safer approach compared to traditional sclerotherapy, particularly for more challenging varicose vein presentations․
UGFS vs․ Endovenous Laser Ablation (EVLA)
Both Ultrasound-Guided Foam Sclerotherapy (UGFS) and Endovenous Laser Ablation (EVLA) are effective treatments for varicose veins, but they differ significantly in their approach․ EVLA utilizes laser energy to heat and close the affected vein, requiring local anesthesia and a more invasive procedure․ UGFS, conversely, involves injecting a sclerosing agent into the vein under ultrasound guidance, causing it to collapse and eventually be reabsorbed by the body․
EVLA is often favored for larger, more tortuous veins, while UGFS is particularly well-suited for smaller varicose veins and those closer to the skin’s surface․ UGFS generally involves less post-procedural pain and a quicker recovery time compared to EVLA․
The choice between the two depends on factors like vein size, location, patient preference, and the physician’s expertise․ Both procedures benefit from ultrasound guidance for accurate targeting and improved outcomes, offering viable solutions for venous insufficiency․

Current NICE Guidance on UGFS (HealthTech Guidance 301)
The National Institute for Health and Care Excellence (NICE), through HealthTech Guidance 301 (previously Interventional Procedures Guidance 440), has evaluated ultrasound-guided foam sclerotherapy (UGFS) for varicose veins․ NICE confirms UGFS as a useful treatment option, particularly for limited and smaller varicose veins, acknowledging its relatively safe profile when performed correctly․
The guidance emphasizes the importance of adequate patient selection and informed consent, ensuring individuals are fully aware of potential risks and benefits․ While the recommendations remain unchanged from the previous guidance, the migration to HealthTech Guidance 301 signifies ongoing review and standardization;
NICE stresses that clinicians should warn patients about the small, but possible, complications associated with UGFS․ The guidance supports its use within established clinical protocols, contributing to consistent and high-quality care for those suffering from venous insufficiency․